Degree of Al-Si order in K-feldspar and its effect on K-feldspar’s dissolution
the effect of Al-Si ordering on K-feldspar’s dissolution
DOI:
https://doi.org/10.13133/2239-1002/17479Abstract
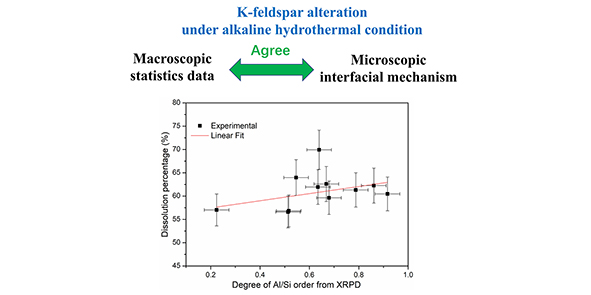
Dissolution or alteration of feldspars is controlled by many factors such as the crystal structure (e.g., Al/Si ordering), temperature, pH, surface area, organic acids, chemical affinity, and precipitation of secondary minerals. The effects of Al/Si ordering (i.e., the degree of Al/Si order) on the percentage of feldspar dissolution (i.e., the extent of feldspar alteration) have not been studied. In this study, the degree of Al/Si order of different types of K-feldspars (eleven specimens including one sanidine sample, five orthoclase samples and five microcline samples) were investigated and determined by both X-ray powder diffraction (XRPD) and Fourier transform infrared spectra (FTIR) method. Further dissolution experiments of K-feldspar samples were operated under the hydrothermal condition for exploring the effects of Al/Si ordering on their dissolution. The degree of Al/Si order between XRPD and FTIR shows a good positive correlation (Pearson’s R =0.94726). The degree of Al/Si order is slightly positively correlated with the percentage of K-feldspars dissolution, and this presents a useful guide for people who are going to utilize K-feldspar as the fertilizer using a hydrothermal alkaline method and try to pick up those samples including orthoclase or microcline from an economic view point.
Published
Issue
Section
License
Copyright (c) 2021 Periodico di Mineralogia

This work is licensed under a Creative Commons Attribution 4.0 International License.

